1. Principle of laser generation
The atomic structure is like a small solar system, with the atomic nucleus in the middle. The electrons are constantly rotating around the atomic nucleus, and the atomic nucleus is also constantly rotating.

The nucleus is composed of protons and neutrons. Protons are positively charged and neutrons are uncharged. The number of positive charges carried by the entire nucleus is equal to the number of negative charges carried by the entire electrons, so generally atoms are neutral to the outside world.
As far as the mass of an atom is concerned, the nucleus concentrates most of the mass of the atom, and the mass occupied by all electrons is very small. In the atomic structure, the nucleus only occupies a small space. The electrons rotate around the nucleus, and the electrons have a much larger space for activity.
Atoms have “internal energy”, which consists of two parts: one is that the electrons have a orbiting speed and a certain kinetic energy; the other is that there is a distance between the negatively charged electrons and the positively charged nucleus, and there is A certain amount of potential energy. The sum of the kinetic energy and potential energy of all electrons is the energy of the entire atom, which is called the internal energy of the atom.
All electrons rotate around the nucleus; sometimes closer to the nucleus, the energy of these electrons is smaller; sometimes further away from the nucleus, the energy of these electrons is larger; according to the probability of occurrence, people divide the electron layer into different ” “Energy Level”; On a certain “Energy Level”, there may be multiple electrons orbiting frequently, and each electron does not have a fixed orbit, but these electrons all have the same level of energy; “Energy Levels” are isolated from each other. Yes, they are isolated according to energy levels. The concept of “energy level” not only divides electrons into levels according to energy, but also divides the orbiting space of electrons into multiple levels. In short, an atom may have multiple energy levels, and different energy levels correspond to different energies; some electrons orbit at a “low energy level” and some electrons orbit at a “high energy level”.
Nowadays, middle school physics books have clearly marked the structural characteristics of certain atoms, the rules of electron distribution in each electron layer, and the number of electrons at different energy levels.
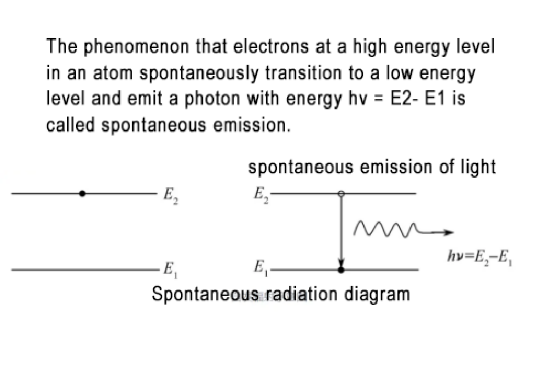
In an atomic system, electrons basically move in layers, with some atoms at high energy levels and some at low energy levels; because atoms are always affected by the external environment (temperature, electricity, magnetism), high-energy level electrons are unstable and will Spontaneous transition to a low energy level, its effect may be absorbed, or it may produce special excitation effects and cause “spontaneous emission”. Therefore, in the atomic system, when high-energy level electrons transition to low-energy levels, there will be two manifestations: “spontaneous emission” and “stimulated emission”.
Spontaneous radiation, electrons in high-energy states are unstable and, affected by the external environment (temperature, electricity, magnetism), spontaneously migrate to low-energy states, and excess energy is radiated in the form of photons. The characteristic of this kind of radiation is that the transition of each electron is carried out independently and is random. The photon states of spontaneous emission of different electrons are different. The spontaneous emission of light is in an “incoherent” state and has scattered directions. However, spontaneous radiation has the characteristics of the atoms themselves, and the spectra of spontaneous radiation of different atoms are different. Speaking of this, it reminds people of a basic knowledge in physics, “Any object has the ability to radiate heat, and the object has the ability to continuously absorb and emit electromagnetic waves. The electromagnetic waves radiated by heat have a certain spectrum distribution. This spectrum The distribution is related to the properties of the object itself and its temperature.” Therefore, the reason for the existence of thermal radiation is the spontaneous emission of atoms.
In stimulated emission, high-energy level electrons transition to a low-energy level under the “stimulation” or “induction” of “photons suitable for the conditions” and radiate a photon of the same frequency as the incident photon. The biggest feature of stimulated radiation is that the photons generated by stimulated radiation have exactly the same state as the incident photons that generate stimulated radiation. They are in a “coherent” state. They have the same frequency and the same direction, and it is completely impossible to distinguish the two. differences among those. In this way, one photon becomes two identical photons through one stimulated emission. This means that the light is intensified, or “amplified”.
Now let’s analyze again, what conditions are needed in order to obtain more and more frequent stimulated radiation?
Under normal circumstances, the number of electrons in high energy levels is always less than the number of electrons in low energy levels. If you want atoms to produce stimulated radiation, you want to increase the number of electrons in high energy levels, so you need a “pump source”, whose purpose is to stimulate more Too many low-energy level electrons jump to high-energy levels, so the number of high-energy level electrons will be more than the number of low-energy level electrons, and a “particle number reversal” will occur. Too many high-energy level electrons can only stay for a very short time. Time will jump to a lower energy level, so the possibility of stimulated emission of radiation will increase.
Of course, the “pump source” is set for different atoms. It makes the electrons “resonate” and allow more low-energy level electrons to jump to high-energy levels. Readers can basically understand, what is laser? How is laser produced? Laser is “light radiation” that is “excited” by the atoms of an object under the action of a specific “pump source”. This is laser.
Post time: May-27-2024
















